The Mahata Group is currently not accepting enquiries from prospective students.
Biography
Bidesh Mahata obtained his PhD in Biochemistry from the University of Calcutta, India, in 2008. In 2009, he moved to the University of Dundee, UK, for his postdoctoral research with Dr Dimitris Xirodimas. In 2011, he joined Teichmann Laboratory, MRC-Laboratory of Molecular Biology, Cambridge, UK, as an investigator scientist. There he discovered that T helper 2 lymphocytes upregulate Cyp11a1 expression, the enzyme that catalyses the first and rate-limiting step of steroid biosynthesis, and produce steroid hormones, possibly to resolve immune reactions and restore homeostasis. Fascinated by that discovery, he went on to develop discovery tools to study immune cell-mediated steroidogenesis. In 2013, the Teichmann Laboratory moved to the EMBL-EBI and Wellcome Sanger Institute, Wellcome Genome Campus, Hinxton, Cambridge, UK. There he took initiatives to generate a novel Cyp11a1-mCherry reporter mouse line to track steroidogenic cells and a conditional knockout mouse line to ablate steroidogenesis cell type specifically. Using these discovery tools and with outstanding support from the Teichmann Laboratory (particularly in the area of single-cell genomics), he discovered that tumour infiltrating T cells, mast cells and basophils are steroidogenic and these steroidogenic immune cells are involved in anti-tumour immunosuppression. In 2019, he joined the Department of Pathology, University of Cambridge, UK, to lead a research team focussed on immune cell steroidogenesis.
Research
Immune cell-mediated steroidogenesis
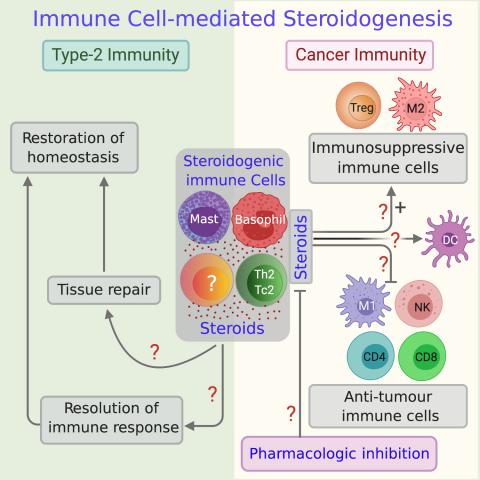
We discovered that immune cells can synthesise and secrete steroid hormones to regulate immune cell function. At present we know very little about the physiological and pathological role of this immune cell-mediated steroid biosynthesis. Therefore, we are curious to know the cause, mechanism and consequence of this immune cell steroidogenesis. We are committed to exploiting this knowledge to improve human health and combat disease.
Our initial studies suggest that immune cell-mediated steroid biosynthesis is involved in type-2 immune responses such as helminth infection, allergies and allergic asthma, pregnancy and many solid tumours. We hypothesise (1) immune cell steroidogenesis pathway is required to resolve immune response and restore tissue homeostasis in helminth infections (2) the pathway is mutated in allergies and allergic asthma, and (3) in solid tumours, steroid-producing immune cells are associated with anti-tumour immunosuppression and immune evasion.
We prioritised to study the role of steroidogenic immune cells in regulating anti-tumour immunity, and aim to develop a novel immunotherapeutic strategy against cancer. We undertake multidisciplinary approaches to answer our questions of why, how and when immune cells produce steroids.
Group members
Dr Hosni Hussein (Postdoctoral research associate): Investigating the role of immune cell steroidogenesis in regulation of inflammation and restoration of tissue homeostasis. Hosni obtained his PhD in Microbiology and Immunology (2018) from the East Carolina University, Greenville, NC, US. Before joining us, he worked at the Harvard Medical School, Massachusetts, USA as a postdoctoral researcher.
Miss Clara Veiga-Villauriz (PhD student): Investigating the role of extra-glandular steroidogenesis and steroid signalling in cancer metastasis. Before joining us, she obtained her MSc in Integrated Immunology (2022) from the University of Oxford.
Mr Qiuchen Zhao (CRUK CC MRes/PhD student): Investigating the role of immune cell-mediated steroidogenesis and steroid signalling in the tumour microenvironment of breast cancer and glioblastoma. Before joining us, he completed his Masters of Research (MRes) training from the Department of Oncology, University of Cambridge.
Mr Soura Chakraborty (Cambridge Trust International PhD student): Investigating the role and regulation of local steroidogenesis and steroid signalling in cancer inflammation and immunity. He is committed to determine the role of steroids signalling in NK cell function. Before joining us, he obtained his MSc in Biotechnology degree (2020) from the Jawaharlal Nehru University.
Dr Sanu K. Shaji (Postdoctoral research associate): Investigating the role of steroidogenic immune cells in the regulation of inflammation-driven cancer progression and targeting immune cell steroidogenesis pathway to reinstate anti-tumour immunity. Sanu did his PhD in Biotechnology (2021) from the School of Biotechnology, Amrita Vishwa Vidyapeetham, Kerala, India. Before joining us, he was a short-term postdoctoral fellow at the Norwich Medical School, University of East Anglia, Norwich, UK, supported by the University's Global Talent Search Fellowship.
Dr Jhuma Pramanik (Postdoctoral research associate): Investigating the regulation of immune cell-mediated steroidogenesis and its role in immune cell function. A major aim of her research is to determine the cell-intrinsic role of immune cell steroids. She did her PhD (Biochemistry) from the Jadavpur University, Kolkata, India. Before joining Mahata lab, she worked with Dr Jon Audhya, University of UW-Madison (2008-2009), Dr Sarah Teichmann, Welcome Sanger Institute (2013-2018), and Dr Virginia Pedicord, University of Cambridge (2018-2019) as a postdoctoral research scientist.
Mr Christopher Ward (PhD Student): Studying the role of glucocorticoid signalling in T lymphocytes function during anti-tumour immune responses, and exploiting that knowledge to develop novel therapeutic approaches (preclinical, experimental, proof of the principle studies) against cancer. Before joining us, he worked in the Buczacki Lab, Wellcome-MRC Cambridge Stem Cell Institute (now relocated to the Oxford University), and Clatworthy Lab, University of Cambridge. Chris obtained his M.Sc. & Diploma of Imperial College, Immunology from the Imperial College London (2016-2017).
Lab Alumni
Dr Rongrong Zhao (Clinician scientist, visiting, 2023): Investigated the role of immune cell steroidogenesis in cancer. At present, she is an oncologist and attending doctor at the Department of Medical Oncology, Shandong Cancer Hospital & Institute.
Dr Md Abdul Alim (SSMF Postdoctoral Research Fellow): Investigated the role of steroid-producing mast cell in B16F10 lung metastasis. He is an immunologist specialised in the area of inflammation and mast cell biology.
Mr Jack Chapman (Part II student, 2023): Investigated the role of StarD4 in regulating T helper (Th2) differentiation and Th2-mediated steroid biosynthesis.
Ms Ananyaa Jain (Part II Student, 2022): Investigated the role of StarD4 in the regulation of mitochondrial cholesterol import in T helper 2 lymphocytes.
Ms Megan Zaman (MPhil Student, 2021): Undertaking a structure-based drug-designing approach, she discovered new inhibitors of the immune cell steroidogenesis pathway.
Ms Beth Brown (Part II Student, 2021): Identified new inhibitors of CYP11A1 by virtual screening.
Collaborators
Professor Sir David Klenerman, University of Cambridge, UK
Professor Klaus Okkenhaug, University of Cambridge, UK
Professor Nicholas Coleman, University of Cambridge, UK
Professor Carlos Caldas, CRUK Cambridge Institute, UK
Professor Wiebke Arlt, University of Birmingham, UK
Dr Amit Roshan, CRUK Cambridge Institute, UK
Dr Amit Agrawal, Cambridge University Hospitals NHS Foundation Trust, UK
Dr Rahul Roychoudhuri, University of Cambridge, UK
Professor William Griffiths, Swansea University, UK
Professor Ruth Andrew and Dr Natalie Homer, University of Edinburgh, UK
Dr Simon Buczacki, University of Oxford, UK
Dr Abhik Mukhopadhyay, Cambridge Crystallographic Data Centre, UK
Dr Xiuwei Zhang, Georgia Institute of Technology, USA
Dr Mingfu Shao, Pennsylvania State University, USA
Key publications
- Zhao, Q., Pramanik, J., Lu, Y., Homer, N. Z. M., Imianowski, C. J., Zhang, B., Iqbal, M., Shaji, S.K., Conway-Morris, A., Roychoudhuri, R., Okkenhaug, K., Qiu, P*. and Mahata, B*. (2025). Perturbing local steroidogenesis to improve breast cancer immunity. Nat Commun 16(1)3945.
- Pramanik, J., Shaji, S. K., Zaman, M., Brown, B., Zhang, B., Yamashita-Kanemaru, Y., Okkenhaug, K., Roychoudhuri, R., Mukhopadhyay, A.*, and Mahata, B.* (2025). Drug repurposing study identified Posaconazole as an indu
- Zhcer of anti-tumour immunity. iScience 28(5)112488.
- Sandor, L.F., Huh, J.B., Benko, P., Hiraga, T., Poliska, S., Dobo-Nagy, C., Simpson, J.P., Homer, N.Z.M., Mahata, B., and Gyori, D.S. (2024). De novo steroidogenesis in tumor cells drives bone metastasis and osteoclastogenesis. Cell Rep 43, 113936.
- Sivakanthan, T., Tanner, J., Mahata, B., and Agrawal, A. (2024). Investigating the role of tumour-to-skin proximity in predicting nodal metastasis in breast cancer. Breast Cancer Res Treat 205, 109-116.
- Roy, S., Roy, S., Mahata, B., Pramanik, J., Hennrich, M.L., Gavin, A.C., and Teichmann, S.A. (2023). CLICK-chemoproteomics and molecular dynamics simulation reveals pregnenolone targets and their binding conformations in Th2 cells. Front Immunol 14, 1229703.
- Roy, S., Sipthorp, J., Mahata, B., Pramanik, J., Hennrich, M.L., Gavin, A.C., Ley, S.V., and Teichmann, S.A. (2021). CLICK-enabledanalogues reveal pregnenolone interactomes in cancer and immune cells. iScience 24, 102485
- Pramanik, J., Zhao, Q., Homer, N.Z.M., Kanemaru, Y., Hussein, H., Chakraborty, S., Roychoudhuri, R., Okkenhaug, K., and Mahata, B.* (2025) Primitive steroidogenesis in mast cells: A novel regulatory mechanism for mast cell function. (bioRxiv DOI: 10.1101/2025.02.05.636621)
- Banik, D., Ward, C.J., Zhang, Z., Heraghty, D., Suresh, P., Li, B., Kedia, S., Davis, S., James Roy, J., Chapman, M., Mahata, B.*, and Klenerman, D.* (2024). Hyaluronidase unlocks sequestered CEACAM5 and improves CAR T-cell therapy in colorectal cancer. (bioRxiv DOI: 10.1101/2024.08.29.610222)
- Ward, C.J., Chakraborty, S., Shaji, S.K., Veiga-Villauriz, C., Zhao, Q., Chen, X. and Mahata, B.* (2025) Glucocorticoid receptor and RUNX transcription factors cooperatively drive CD8 T cells dysfunction in human cancer. (bioRxiv DOI: 10.1101/2025.04.30.651472)
- Hussein, H.A.M.*, Shaji, S.K., Veiga-Villauriz, C., Chakraborty, S., Pramanik, J., Ali, F.A.Z., Yuan, J., Khanfar, E., Makuyana, N., Ali, Y.M., Zhao, Q., and Mahata, B.* (2025) Immune cell de novo steroidogenesis regulates inflammation resolution and recovery in acute lung injury. (bioRxiv DOI: 10.1101/2025.08.29.673039)
- Chakraborty S, Pramanik J, and Mahata B. (2021) Revisiting steroidogenesis and its role in immune regulation with the advanced tools and technologies. Genes & Immunity. https://doi.org/10.1038/s41435-021-00139-3
- Mahata B*, Pramanik J, van der Weyden L, Polanski K, Kar G, Riedel A, Fonseca NA, Kundu K, Campos LS, Ryder E, Duddy G, Walczak I, Davidson S, Okkenhaug K, Adams DJ, Shields JD* and Teichmann SA*. (2020) Tumors induce de novo steroid biosynthesis in T cells to evade immunity. Nature Communications https://www.nature.com/articles/s41467-020-17339-6
- Pramanik J, Chen X, Kar G, Henriksson J, Gomes T, Park J, Natarajan K, Meyer KB, Miao Z, McKenzie AN, Mahata B*, and Teichmann SA*. (2018) Genome-wide analyses reveal the IRE1a-XBP1 pathway promotes T helper cell differentiation by resolving secretory stress and accelerating proliferation. Genome Medicine 10:76
- Mahata B*, Zhang X, Kolodziejczyk AA, Proserpio V, Haim-Vilmovsky L, Taylor AE, Hebenstreit D, Dingler FA, Moignard V, Göttgens B, Arlt W, McKenzie AN, Teichmann SA*. (2014) Single-cell RNA sequencing reveals T helper cells synthesizing steroids de novo to contribute to immune homeostasis. Cell Reports 7: 1130-1142.*co-corresponding author
Publications
- Zhao, Q., Pramanik, J., Lu, Y., Homer, N. Z. M., Imianowski, C. J., Zhang, B., Iqbal, M., Shaji, S.K., Conway-Morris, A., Roychoudhuri, R., Okkenhaug, K., Qiu, P*. and Mahata, B*. (2025). Perturbing local steroidogenesis to improve breast cancer immunity. Nat Commun 16(1)3945.
- Pramanik, J., Shaji, S. K., Zaman, M., Brown, B., Zhang, B., Yamashita-Kanemaru, Y., Okkenhaug, K., Roychoudhuri, R., Mukhopadhyay, A.*, and Mahata, B.* (2025). Drug repurposing study identified Posaconazole as an indu
- Zhcer of anti-tumour immunity. iScience 28(5)112488.
- Sandor, L.F., Huh, J.B., Benko, P., Hiraga, T., Poliska, S., Dobo-Nagy, C., Simpson, J.P., Homer, N.Z.M., Mahata, B., and Gyori, D.S. (2024). De novo steroidogenesis in tumor cells drives bone metastasis and osteoclastogenesis. Cell Rep 43, 113936.
- Sivakanthan, T., Tanner, J., Mahata, B., and Agrawal, A. (2024). Investigating the role of tumour-to-skin proximity in predicting nodal metastasis in breast cancer. Breast Cancer Res Treat 205, 109-116.
- Roy, S., Roy, S., Mahata, B., Pramanik, J., Hennrich, M.L., Gavin, A.C., and Teichmann, S.A. (2023). CLICK-chemoproteomics and molecular dynamics simulation reveals pregnenolone targets and their binding conformations in Th2 cells. Front Immunol 14, 1229703.
- Roy, S., Sipthorp, J., Mahata, B., Pramanik, J., Hennrich, M.L., Gavin, A.C., Ley, S.V., and Teichmann, S.A. (2021). CLICK-enabledanalogues reveal pregnenolone interactomes in cancer and immune cells. iScience 24, 102485
- Pramanik, J., Zhao, Q., Homer, N.Z.M., Kanemaru, Y., Hussein, H., Chakraborty, S., Roychoudhuri, R., Okkenhaug, K., and Mahata, B.* (2025) Primitive steroidogenesis in mast cells: A novel regulatory mechanism for mast cell function. (bioRxiv DOI: 10.1101/2025.02.05.636621)
- Banik, D., Ward, C.J., Zhang, Z., Heraghty, D., Suresh, P., Li, B., Kedia, S., Davis, S., James Roy, J., Chapman, M., Mahata, B.*, and Klenerman, D.* (2024). Hyaluronidase unlocks sequestered CEACAM5 and improves CAR T-cell therapy in colorectal cancer. (bioRxiv DOI: 10.1101/2024.08.29.610222)
- Ward, C.J., Chakraborty, S., Shaji, S.K., Veiga-Villauriz, C., Zhao, Q., Chen, X. and Mahata, B.* (2025) Glucocorticoid receptor and RUNX transcription factors cooperatively drive CD8 T cells dysfunction in human cancer. (bioRxiv DOI: 10.1101/2025.04.30.651472)
- Hussein, H.A.M.*, Shaji, S.K., Veiga-Villauriz, C., Chakraborty, S., Pramanik, J., Ali, F.A.Z., Yuan, J., Khanfar, E., Makuyana, N., Ali, Y.M., Zhao, Q., and Mahata, B.* (2025) Immune cell de novo steroidogenesis regulates inflammation resolution and recovery in acute lung injury. (bioRxiv DOI: 10.1101/2025.08.29.673039)
- Chakraborty S, Pramanik J, and Mahata B. (2021) Revisiting steroidogenesis and its role in immune regulation with the advanced tools and technologies. Genes & Immunity. https://doi.org/10.1038/s41435-021-00139-3
- Mahata B*, Pramanik J, van der Weyden L, Polanski K, Kar G, Riedel A, Fonseca NA, Kundu K, Campos LS, Ryder E, Duddy G, Walczak I, Davidson S, Okkenhaug K, Adams DJ, Shields JD* and Teichmann SA*. (2020) Tumors induce de novo steroid biosynthesis in T cells to evade immunity. Nature Communications https://www.nature.com/articles/s41467-020-17339-6
- Pramanik J, Chen X, Kar G, Henriksson J, Gomes T, Park J, Natarajan K, Meyer KB, Miao Z, McKenzie AN, Mahata B*, and Teichmann SA*. (2018) Genome-wide analyses reveal the IRE1a-XBP1 pathway promotes T helper cell differentiation by resolving secretory stress and accelerating proliferation. Genome Medicine 10:76
- Mahata B*, Zhang X, Kolodziejczyk AA, Proserpio V, Haim-Vilmovsky L, Taylor AE, Hebenstreit D, Dingler FA, Moignard V, Göttgens B, Arlt W, McKenzie AN, Teichmann SA*. (2014) Single-cell RNA sequencing reveals T helper cells synthesizing steroids de novo to contribute to immune homeostasis. Cell Reports 7: 1130-1142.*co-corresponding author