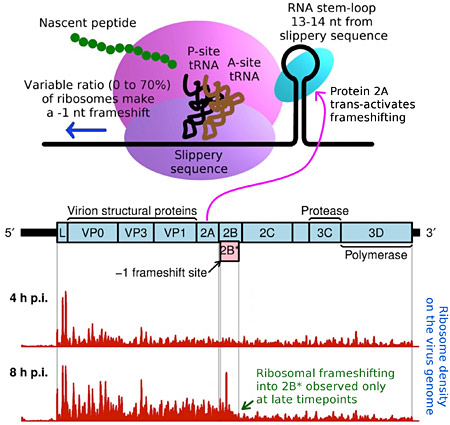
A new study from the Firth and Brierley groups has just been published in Nature Communications. Programmed −1 ribosomal frameshifting is a mechanism of gene expression, whereby specific signals within messenger RNAs (mRNAs) direct a proportion of translating ribosomes to shift −1 nt and continue translating in the new reading frame. Such frameshifting is normally stimulated by RNA secondary structure within the mRNA, resulting in a fixed ratio of frameshift and non-frameshift protein products. In this study, researchers found that frameshifting in a model RNA virus – encephalomyocarditis virus – is trans-activated by one of the virus proteins. As a result, the efficiency of frameshifting increases from 0% to 70% as the amount of viral protein builds up in an infected cell, leading to temporal regulation of the expression levels of the viral structural and enzymatic proteins. First author Dr Sawsan Napthine mapped out the molecular interactions underlying this novel mechanisms of regulating virus gene expression. Other experiments were performed by Drs Leanne Finch, Joshua Jones and Roger Ling.
https://www.nature.com/articles/ncomms15582
Napthine S, Ling R, Finch LK, Jones JD, Bell S, Brierley I, Firth AE (2017) Nat Commun, Jun 8, 8:15582.

