Currently not taking Postgraduate Students.
Research
Our group focuses on the roles that endosomes and exosomes play in health and disease.
Endosomes are intracellular membrane-bound organelles that have several important roles. Endosomes regulate the trafficking of proteins and lipids between several pathways including the endocytic, secretory, recycling and degradative. As endosomes mature, proteins on the limiting membrane of endosomes can be sorted into domains that tubulate or vesiculate towards the cytosol for recycling. Conversely, proteins can be sorted to domains that subsequently invaginate inwards, away from the cytosol, where they form intralumenal vesicles (ILVs). Once an endosome has acquired ILVs, it becomes termed a late endosome, or multivesicular body (MVB). Trafficking a protein to an ILV has several functions; firstly it removes any signalling capacity of these proteins from interactions with the cytosol (this is especially important for downregulation of activated growth factor receptors), and secondly it packages proteins into vesicles that can be degraded or expelled from the cell.
MVBs have several potential fates. Fusion of an MVB with a lysosome forms a hybrid organelle, an endolysosome, where ILVs and their contents are degraded and recycled. Another fate of MVBs is that they may fuse with the plasma membrane where their ILVs are released into the extracellular milieu - these ILVs now become termed 'exosomes'.
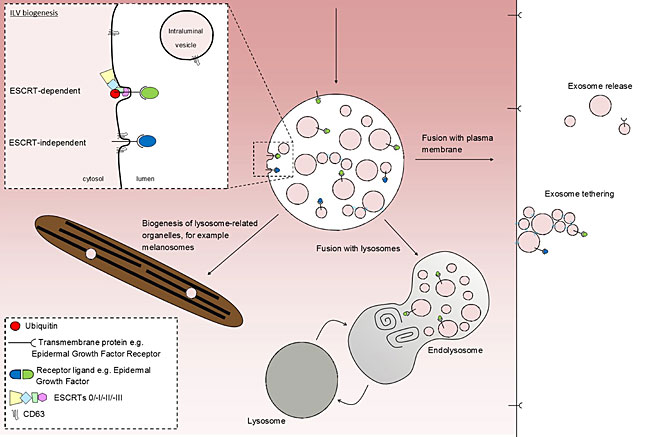
Exosomes are suggested to be involved in intercellular (between different cells) communication and have been implicated in a number of physiological and pathological processes, including antigen presentation, cancer and neurodegeneration. However, the basic cell biology which underlies their biology is relatively poorly understood. Our lab aims to identify the molecular machinery which governs the behaviour of endosomes and exosomes.
We have recently shown that in mammalian cells exosomes can be tethered to the plasma membrane of cells following their release. This tethering is dependent upon a protein called tetherin, which plays a similar role in the retention of a number of virions. This highlights the possibility that exosomes have roles in short-range communication, in addition to longer-range communications. Our group will focus on understanding how and why exosomes are retained by this tethering process.
Current areas of interest:
* Molecular mechanisms of exosome tethering
* The role of exosomes in MHC-II presentation
* PD-L1 trafficking
* Exosome transfer, uptake and delivery
* MT1-MMP trafficking
Publications
Jackson HK, Long HM, Yam-Puc JC, Palmulli R, Haigh TA, Gerber PP, Lee JS, Matheson NJ, Young L, Trowsdale J, Lo M, Taylor GS, Thaventhiran JE, Edgar JR.
J Extracell Vesicles. 2024 Feb;13(2):e12412. doi: 10.1002/jev2.12412.
Tetherin antagonism by SARS-CoV-2 ORF3a and spike protein enhances virus release.
Stewart H, Palmulli R, Johansen KH, McGovern N, Shehata OM, Carnell GW, Jackson HK, Lee JS, Brown JC, Burgoyne T, Heeney JL, Okkenhaug K, Firth AE, Peden AA, Edgar JR.
EMBO Rep. 2023 Dec 6;24(12):e57224. doi: 10.15252/embr.202357224. Epub 2023 Oct 11.
Tetherin is an exosomal tether.
Edgar JR, Manna PT, Nishimura S, Banting G, Robinson MS.
Elife. 2016 Sep 22;5. pii: e17180. doi: 10.7554/eLife.17180.
Q&A: What are exosomes, exactly?
Edgar JR.
BMC Biol. 2016 Jun 13;14:46. doi: 10.1186/s12915-016-0268-z.
Edgar JR, Willén K, Gouras GK, Futter CE.
J Cell Sci. 2015 Jul 15;128(14):2520-8. doi: 10.1242/jcs.170233. Epub 2015 May 22.
Hrs- and CD63-dependent competing mechanisms make different sized endosomal intraluminal vesicles.
Edgar JR, Eden ER, Futter CE.
Traffic. 2014 Feb;15(2):197-211. doi: 10.1111/tra.12139. Epub 2014 Jan 8.
Hirst J, Edgar JR, Esteves T, Darios F, Madeo M, Chang J, Roda RH, Dürr A, Anheim M, Gellera C, Li J, Züchner S, Mariotti C, Stevanin G, Blackstone C, Kruer MC, Robinson MS.
Hum Mol Genet. 2015 Sep 1;24(17):4984-96. doi: 10.1093/hmg/ddv220. Epub 2015 Jun 17.
For a complete list of publications, please click here
Teaching and Supervisions
MHC Class II Antigen Presentation - Part II, Immunology
Anti-viral Restriction Factors - Part II, Virology
Membrane Traffic (4 lectures) - Part 1B, Cell and Developmental Biology
MPhil skills session - microscopy
Lab members:
Dr Yağmur Yıldızhan (PDRA)
Mr Adam Bourke (PhD student)
Ms Trinity Eales (MPhil student)
Mr Jin Seok Lee (PhD student, co-supervised with Klaus Okkenhaug)
Previous lab members:
Dr Roberta Palmulli (PDRA)
Dr Hannah Jackson (PDRA)
Miss Anna Germon (PhD student)
Dr Panchali Kanvatirth (PDRA)
Funding
Wellcome Trust
The Royal Society
The Wellcome Trust Institutional Strategic Support Fund (ISSF)
Exosis inc, Florida (2022 - 2023)
Other Professional Activities
Molecules and Cells - Theme Lead (2024 - )


