Submitted by Administrator on Thu, 19/03/2015 - 10:17
Following lactation, milk-producing mammary epithelial cells are removed by an exquisitely controlled process of cell death. Having previously demonstrated that the transcription factor Stat3 regulates a lysosomal-mediated programme of cell death (LM-PCD) during mammary gland involution, Tim Sargeant and Bethan Lloyd-Lewis in Christine Watson’s lab report this week in Nature Cell Biology the mechanism that controls the release of lysosomal cathepsins to initiate cell death.
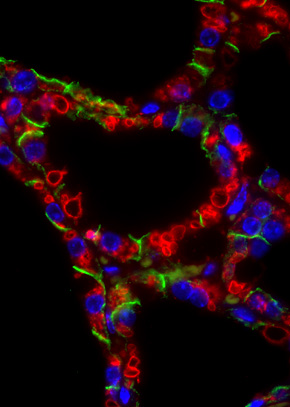 As shown in the striking image of red-rimmed structures in involuting mammary tissue, Stat3 regulates the formation of large lysosomal vacuoles that contain triglyceride which is found in milk. It was demonstrated that milk fat globules (MFGs) are toxic to epithelial cells and that, when applied to purified lysosomes, the MFG-derived fatty acid, oleic acid, potently induces lysosomal leakiness. Additionally, uptake of secreted MFGs is diminished in Stat3-ablated mammary glands while loss of the phagocytosis bridging molecule MFG-E8 results in reduced leakage of cathepsins in vivo. The Watson lab propose that Stat3 regulates LM-PCD in mouse mammary gland by switching cellular function from secretion of milk to uptake of MFGs. Consequently, lysosomal vesicle membranes become perturbed by high levels of free fatty acids resulting in controlled leakage of cathepsins. This culminates in cell death. Since cancer cells are often refractory to apoptosis and frequently harbour constitutively active Stat3, understanding the mechanism of LM-PCD and the critical role of Stat3 in this process, will provide new approaches to cancer therapy.
As shown in the striking image of red-rimmed structures in involuting mammary tissue, Stat3 regulates the formation of large lysosomal vacuoles that contain triglyceride which is found in milk. It was demonstrated that milk fat globules (MFGs) are toxic to epithelial cells and that, when applied to purified lysosomes, the MFG-derived fatty acid, oleic acid, potently induces lysosomal leakiness. Additionally, uptake of secreted MFGs is diminished in Stat3-ablated mammary glands while loss of the phagocytosis bridging molecule MFG-E8 results in reduced leakage of cathepsins in vivo. The Watson lab propose that Stat3 regulates LM-PCD in mouse mammary gland by switching cellular function from secretion of milk to uptake of MFGs. Consequently, lysosomal vesicle membranes become perturbed by high levels of free fatty acids resulting in controlled leakage of cathepsins. This culminates in cell death. Since cancer cells are often refractory to apoptosis and frequently harbour constitutively active Stat3, understanding the mechanism of LM-PCD and the critical role of Stat3 in this process, will provide new approaches to cancer therapy.
Stat3 controls cell death during mammary gland involution by regulating uptake of milk fat globules and lysosomal membrane permeabilization.
Sargeant TJ, Lloyd-Lewis B, Resemann HK, Ramos-Montoya A, Skepper J, Watson CJ. Nat Cell Biol. 2014 Oct 5. doi: 10.1038/ncb3043.
