Biography
I was awarded my BSc cum laude and with General Honors, from the University of Miami, majoring in Microbiology & Immunology, with a double minor in Chemistry and Biology. I was awarded my PhD and double Masters degrees from Columbia University, Department of Microbiology & Immunology in New York City. I worked with Professor David Shore on transcriptional silency and heterochromatin assembly in the budding yeast, Saccharomyces cerevisiase.
After being granted a Fellowship from the Imperial Cancer Research Fund (now the Francis Crick Institute), I moved to London to do my post-doctoral research on KSHV-encoded cyclins with Dr Nic Jones and cell cycle regulation with Dr Gordon Peters. After discovering the importance of ubiquitin ligases for G1 phase kinases in KSHV-driven malignancies, I was funded by the Association for International Cancer Research to work with Professor Chris Boshoff at the Wolfson Institute for Biomedical Research at University College London.
I was awarded a Research Fellowship to join the Department of Pathology as an early career Group Leader at the University of Cambridge and am now a Professor in Cellular and Molecular Biology and the Head of the Department. Having been admitted as a Fellow to Clare College, Cambridge I am also a Director of Studies for Pathology and Genetics and the Post-graduate Admissions Tutor.
Research
Research overview
How organisms achieve their intriguing biological complexity with an amazing ability to respond to environmental stress and infections is a fascinating and perplexing question. One strategy is through the action of enzymes that modify proteins to rapidly change protein behaviour and function. Ubiquitin ligases are a family of enzymes my lab investigates which are integral to this. They tag their substrates with a 76 amino acid tag, called ubiquitin. This modification can change a protein’s stability, localisation and trafficking within the cell. Ubiquitin signalling has different consequences in different cell types and under different conditions. My lab studies this signalling in normal and pathological conditions to understand the mechanisms of a wide range of diseases, like Parkinson’s disease, cancer, male sterility and anaemia. We aim to exploit the exquisite specificity of ubiquitin ligase enzymology to invent novel therapeutics.
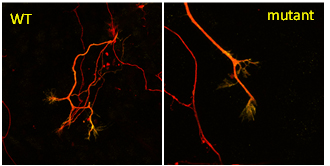
Neuroprotective roles of Fbxo7/PARK15
Parkinson’s disease is the second most common neurodegenerative disease, affecting more than 6 million people worldwide, where the majority of cases are sporadic and approximately 10% of cases are inherited. In 2008, the first case of Parkinsonian pyramidal syndrome was linked to a mutation in FBXO7, and since then many other pathological recessive mutations have been identified. Having shown Fbxo7 acts in a common pathway with the stress-activated kinase, PINK1, and another ubiquitin ligase, Parkin, to enable stress-activated mitophagy, we are using dopaminergic cell and mouse models to identify which cellular pathways will rescue the ensuing neuronal cell death caused by the loss of Fbxo7.
Fbxo7 as a scaffold for the G1 phase cell cycle kinase, Cdk6
During the G1 phase of the cell cycle, the cell integrates extracellular and intracellular signalling and ‘decides’ whether to progress into S phase and initiate DNA synthesis. This transition is safeguarded by the retinoblastoma family of checkpoint proteins, which are inactivated by phosphorylation by the G1 kinases, cyclin D/Cdk4 or cyclin D/Cdk6. More recent descriptions of distinct substrate preferences, restricted expression patterns, and cell-cycle independent roles for Cdk4 and Cdk6 allow us to distinguish between the functions of these kinases.
We discovered Fbxo7 functions as a specific assembly factor for Cdk6 with D-type cyclins, and have described the pathological effects of the loss of its expression on the cell cycle of highly proliferative cell types like erythroblasts and T lymphoblasts. Our goal is to understand whether the loss of Fbxo7’s pathological effects are mediated through its role in regulating Cdk6 control of cell proliferation via pRb or its other substrates and cell cycle-independent functions.
Fbxo7 and PI31 as proteasome regulators
In studying the structural organisation and domains of Fbxo7, we discovered it also contains an FP (Fbxo7-PI31) dimerisation domain that enables its homo-dimerization and hetero-dimerisation with the proteasome regulator, PI31/PSMF1. These proteins share a similar organisational structure; both contain an FP domain and a PRR domain. In cells with specialised morphologies, like neurons, sperm, and erythrocytes, reduced Fbxo7 levels correlates with lower PI31 levels. Multiple global proteomic profiling and interactome studies have demonstrated the association of Fbxo7 and PI31 with each other and the proteasome. Our goal is to understand how Fbxo7 and PI31 coordinate their regulation of the proteasome in cells with specialised morphologies.
F-box proteins in cancer
F-box proteins, including Skp2 and Fbxw7, control the ubiquitin-mediated degradation of key cell cycle regulators, like p27 and cyclin E and Myc. We aim to assess whether when tumours present with mutations in F-box protein genes, whether these can be used as biomarkers for deciding on chemotherapeutic treatments for patients. We collaborate with AstraZeneca in understanding the tumour biology of subsets of breast cancers with regard to their ubiquitin signalling networks.
Targeting ubiquitin ligases and ubiquitinated proteins with novel biotherapeutic approaches
Recent advances in converting small molecule ligands into protein-targeting chimeric (Protacs) molecules demonstrate the power of the ubiquitin-proteasome system to selectively degrade proteins within the cell. Ubiquitin ligases are remarkably specific enzymes that target their substrates via their F-box domain containing subunits. We want to exploit the ability of F-box proteins to bind their substrate to identify protein-protein interacting sites to convert into biological Protacs. We use camelid-derived antibodies known as nanobodies to test and disrupt the interactions between ubiquitin ligases and their oncogenic substrates. Our goal is to define the protein-docking sites for ligands in difficult-to-target oncogenes
Group Members:
Sara Al Rawi, Hanna Bjone, Linda Lin, Sophie Willis, Gudrun Agnarsdottir, Libby Wiseman
Publications
-
J Shen, Z Qiu, Q Wu, G Zhang, R Harris, D Sun, J Rantala, W Barshop, L Zhao, D Lv, K Won, J Wohlschlegel, O Sangfelt, H Laman, J Rich, C Spruck. A FBXO7/EYA2-SCFFBXW7 axis promotes AXL-mediated maintenance of mesenchymal and immune evasion phenotypes of cancer cells. Mol Cell. 2022. Link.
-
R. Harris, S.J. Randle & H Laman. Analysis of the FBXO7 promoter reveals overlapping Pax5 and c-Myb binding sites functioning in B cells. 2021. Biochem Biophys Res Comm. Link
-
Licchesi JDF, Laman H, Ikeda F, Bolanos-Garcia VM. Front Physiol. 2020 Nov 26;11:621053. Link.
-
Mason BJ & Laman H. The FBXL family of F-box proteins: variations on a theme. Open Biol. 2020 Nov;10(11):200319. Link
-
Licchesi JDF, Laman H, Ikeda F, Bolanos-Garcia VM. E3 Ubiquitin Ligases: From Structure to Physiology. Front Physiol. 2020. 26 November 2020 Link
-
Spagnol V, Oliveira CAB, Randle SJ, Passos PMS, Correia CRSTB, Simaroli NB, Oliveira JS, Mevissen TET, Medeiros AC, Gomes MD, Komander D, Laman H, Teixeira FR.The E3 ubiquitin ligase SCF(Fbxo7) mediates proteasomal degradation of UXT isoform 2 (UXT-V2) to inhibit the NF-kappa B signaling pathway. Biochimica et Biophysica Acta. 2020 Sep 30;1865(1):129754. doi: 10.1016/j.bbagen.2020.129754. Link
-
Rathje CC, Randle SJ, Al Rawi S, Skinner BM, Rajamundar A, Nelson DE, Johnson EEP, Bacon J, Vlazak M, Affara NA, Ellis PJ, Laman H.A conserved requirement for Fbxo7 during male germ cell cytoplasmic remodelling. Front Physiol. 2019. Oct 10;10:1278. doi: 10.3389/fphys.2019.01278. Link.
-
Mason B, Flach S, Teixeira, FR, Manzano Garcia R, Rueda OM, Abraham JE, Caldas C, Edwards PAW. Laman H. Fbxl17 is rearranged in breast cancer and loss of its activity leads to increased global O-GlcNAcylation. Cell Mol Life Sci. 2019 Sept 27. Link.
-
SRW Stott†, SJ Randle†, S al Rawi, PA Rowicka, R Harris2, B Mason, J Xia, JW Dalley, RA Barker, H Laman. Loss of FBXO7 results in a Parkinson’s-like dopaminergic degeneration via an RPL23-MDM2-TP53 pathway. J Pathol. 2019 Oct;249(2):241-254. Link.
-
Patel SP, Randle SJ, Gibbs S, Cooke A, Laman H. Opposing effects on the cell cycle of T lymphocytes by Fbxo7 via Cdk6 and p27. Cell Mol Life Sci. 2017 Apr;74(8):1553-1566 . Link.
-
Teixeira FR, Randle SJ, Patel SP, Mevissen TE, Zenkeviciute G, Koide T, Komander D, Laman H. Gsk3β and Tomm20 are substrates of the SCFFbxo7/PARK15 ubiquitin ligase associated with Parkinson's disease. Biochem J. 2016 Oct 15;473(20):3563-3580. Link.