Submitted by Administrator on Thu, 12/12/2013 - 15:17
21 January 2011
Department researchers reveal in PNAS how membrane efflux pumps open to confer multidrug resistance and virulence on bacteria.
Published this month: Pei X-Y, Hinchliffe P, Symmons M, Koronakis E, Hughes C, Koronakis V (2011) Structures of sequential open states in a symmetrical opening transition of the TolC exit duct. Proceedings of the National Academy of Sciences (USA) 108: published online ahead of print January 18, 2011, doi:10.1073/pnas.1012588108
Efflux pumps expel antibiotics and other drugs, so they are important in bacterial multidrug resistance which is a growing threat to clinical treatment of infections by pathogens like Salmonella, E. coli and Pseudomonas. They also export toxins that attack our cells, increasing the ability of the bacteria to cause disease. These ubiquitous pumps must cross two bacterial membranes, inner and outer, and the intervening peripasmic space, they are therefore more complex than drug efflux pumps of human cancer cells and malaria parasites, which have to cross a single membrane. The bacterial pumps are extraordinary biological nanomachines that hoover up drugs or bind toxins via a transporter in the inner membrane, which delivers them to an outer membrane "trash chute" known as the TolC exit duct, with the aid of a third component - the "adaptor" – which connects the two components and opens the TolC exit duct. Professors Vassilis Koronakis and Colin Hughes and colleagues in the Department have spent two decades studying the structure and function of these pumps, assembling the domains of the pump till in 2009 they, with the department’s Oppenheimer Fellow Dr Martyn Symmons, managed to piece together the structure of the whole drug efflux nanomachine (see 2009 Pathology News article: Research reveals new information about antibiotic resistance in bacteria) .
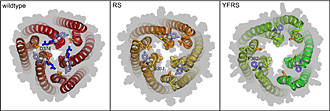
Now these researchers have, with the laboratory expertise of Drs Philip Hinchliffe and Xue-Yuan Pei and Eva Koronakis,
shown how the TolC exit duct entrance opens to allow diverse toxic molecules to be ejected from the cell. TolC is a unique biological structure and looks like a large homotrimeric cannon, its periplasmic entrance where the drugs and toxins are loaded is closed by a network of a-helices and it must be opened to fire the dangerous cargo out of the cell, either endowing resistance to antibiotics or directing toxins at our cells during infection. The Pathology research group solved two high resolution crystal structures of sequential stages in the opening transition, which they induced and stabilised biochemically and measured by defining their electro-conductance when reconstituted in lipid bilayers. The structures at 2.9A show a symmetrical opening mechanism in which TolC helices forming the cannon-shaped channel are relaxed, (in the pump by repacking against helices of the adaptor), to twist and dilate the entrance to its open state. The research reveals further the workings of an exquisite and complex biological nanomachine, and also suggests how one could inhibit these pumps, as the group has shown the TolC entrance can be ligated with inhibitors to prevent its opening.
--------
Koronakis et al (2000) Crystal structure of the bacterial membrane protein TolC central to multidrug efflux and protein export. Nature 405: 914 – 19
Andersen et al (2002). Transition to the open state of the TolC periplasmic tunnel entrance. PNAS 99:11103–8
Higgins et al (2004) Structure of the periplasmic component of a bacterial antibiotic efflux pump. PNAS 101:9994-9
Lobedanz et al (2007) A periplasmic coiled-coil interface underlying TolC recruitment and the assembly of bacterial drug efflux pumps. PNAS 104: 4612-17
Symmons et al (2009) Assembled structure of complete tripartite bacterial multidrug efflux pump. PNAS 106:7173-8
Pei et al (2011) Structures of sequential open states in a symmetrical opening transition of the TolC exit duct. PNAS 108: published online ahead of print January 18, 2011, doi:10.1073/pnas.1012588108
