Submitted by Administrator on Wed, 11/12/2013 - 13:27
7 April 2009
A team led by Professor Vassilis Koronakis in the Department has uncovered an unexpected pathway by which Salmonella manipulates the mamalian host cell to promote bacterial intracellular growth in membrane-bound Salmonella-containing vacuoles (SCVs). Their latest findings are published this week in Cell Host Microbe.
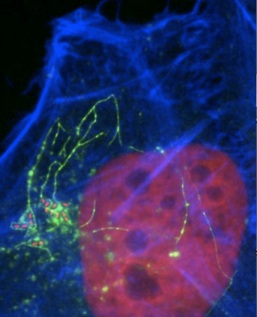 Salmonella are infamous intestinal pathogens that infect humans and animals, causing each year throughout the world about 1.3 billion cases of human food-borne diarrhoea and systemic typhoid fever. These bacteria establish infections by invading host cells, surviving and growing inside them in membrane-bound Salmonella-containing vacuoles (SCVs). Salmonella does this by delivering a cocktail of virulence effector proteins that take control of the cytoskeleton and networks of signalling pathways that regulate key cell activities.
Salmonella are infamous intestinal pathogens that infect humans and animals, causing each year throughout the world about 1.3 billion cases of human food-borne diarrhoea and systemic typhoid fever. These bacteria establish infections by invading host cells, surviving and growing inside them in membrane-bound Salmonella-containing vacuoles (SCVs). Salmonella does this by delivering a cocktail of virulence effector proteins that take control of the cytoskeleton and networks of signalling pathways that regulate key cell activities.
To understand how Salmonella establishes its complex programme of infection, it is essential to identify these host cell targets of the bacterial effector proteins and understand how the targets are manipulated. Such studies also uncover new aspects of mammalian cell biology and present potential new targets to counteract bacterial disease.
Recently, a team led by Professor Vassilis Koronakis in the Department has teased out key features of an unexpected pathway by which Salmonella manipulates the host cellto promote bacterial intracellular growth. They engineered Salmonella to deliver a mutant effector protein (called SptP) in which the effector's active site was disabled to stall enzymatic activity and thus capture its host cell target protein. This approach revealed that the Salmonella SptP effector targets a host ATPase called valosin-containing protein (VCP), which controls assembly of host protein complexes underlying membrane fusion that are central to the host's ability to build intracellular organelles. Using protein biochemistry and fluorescence microscopy of infected cells, Humphreys and colleagues showed that SptP dephosphorylates host cell VCP to subvert regulation of key membrane fusion events, so ensuring the development of a safe, rich intracellular replicative niche for the pathogen.
For more information contact Professor Vassilis Koronakis (vk103@cam.ac.uk). This work was funded by the Wellcome Trust.
